A surprising molecular switch
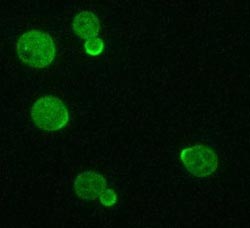
Membrane-bound Cdc42 (shown in green) is a key regulator of cell polarity in yeast. A process guided by filaments concentrates Cdc42 molecules in one spot – the spot where the bud will emerge. Credit: Image: Courtesy of Sarah Smith, Stowers Institute for Medical Research<br>
In a standard biology textbook, cells tend to look more or less the same from all sides. But in real life cells have fronts and backs, tops and bottoms, and they orient many of their structures according to this polarity explaining, for example, why yeast cells bud at one end and not the other.
Over the last few years, Rong Li, Ph.D., and her team at the Stowers Institute for Medical Research have figured out many important details of the basic biochemical mechanism that establishes cell polarity in yeast. Now, using cutting-edge microscopy and advanced mathematics, the team has discovered a surprising new twist. In the March issue of Nature Cell Biology, predoctoral researcher Arupratan Das, Li, and five colleagues report that, in yeast, an enzyme literally flips a switch by moving molecules called phospholipids from the outer to the inner layer of the cell membrane.
What's more, all the molecules involved in this intricate mechanism are found not just in yeast, but in mammalian cells as well. That opens up new avenues for exploring how cell polarity is created in, say, liver cells, and whether or not glitches in the mechanism can contribute to disease. “Cell polarity is critical for the specialized function of the vast majority of cells,” explains Li, who is also a professor in the Department of Molecular and Integrative Physiology at the University of Kansas Medical Center. “Our finding could lead to a lot of clues mechanistically. We're hoping that others will pick up on our discovery and apply it to the cells they study.”
Several decades ago, scientists thought that cell polarity might result from external cues—such as spatially localized chemical signals outside cells that tell the cells how to move and behave. However, researchers have also long noticed that without such localized signals cells can still polarize suggesting that the ability to polarize and bud is innate in the cell.
Li's team uncovered an important clue to the underlying process. Embedded in the cell membrane is a molecule, named Cdc42, that acts as a key regulator. In a non-polarized cell, Cdc42 is randomly distributed around the membrane, like stones randomly set in a brick wall. It also floats around within the cell.
When activated, Cdc42 stimulates the formation of a skeleton of microfilaments that guide free-floating Cdc42 towards the Cdc42 already in the membrane. Some parts of the membrane start out with a bit more Cdc42 than others and the process guided by the microfilaments concentrates the molecule in those areas. Eventually, cells ends up with a high concentration of Cdc42 in one spot—the spot where the bud will emerge.
Of course it isn't that simple. Membrane-bound Cdc42 can diffuse away but can also be pulled out of the membrane by another molecule named Rdi1and then recycled back to the same spot on the membrane. The speed of the recycling determines how big the eventual spot will be, which directly affects the shape of the bud that grows out.
Imagine ants in a ditch, says Li. They keep running out. To keep them there, you pick them up and put them back. If your hands are fast, you can contain them in a very small area. If your hands are slow, the ants spread out more. It's the same in the yeast cell. Faster recycling of Cdc42 results in a smaller spot on the cell; slower recycling causes a bigger spot. In a 2009 paper, Li's team showed that the recycling process can be fast or slow.
So what controls the speed of Cdc42 dissociation and recycling? Li and her colleagues tackled the question by looking for genes that, when knocked out, affect the speed. They found one that slowed things way down. It was a big surprise. “It wasn't something we were envisioning,” says Li. In fact, she wasn't even sure if her team would pursue it. The reason: The gene was for a type of an enzyme known as a lipid flippase, which literally flips lipids from one side of cell membranes to the other. And lipids are “really messy and hard to work with,” she says. Predoctoral researcher Arupratan Das, however, was willing to take it on. “I have to give Das a lot of credit—he's fearless,” says Li.
Using cutting-edge fluorescence fluctuation spectroscopy, Das and his colleagues were able to chart the movement of molecules in live yeast cells, and figure out the role of the flippase. Here's what they found:
Near one end of the Cdc42 molecule is a patch with a net positive electrical charge. The cell membrane has a negative charge. As a result, the positively-charged patch of Cdc42 acts like a piece of Velcro, holding the molecule close to the membrane, allowing a lipid anchor at the end of Cdc42 to be stably inserted in the membrane. To pull out the Cdc42 for recycling, Rdi1 must grab this anchor. “Rdi1 cannot go into the membrane, so it needs the Velcro to be loosened,” Li explains.
That's where the flippase enzyme comes in. The enzyme was known to flip a charge-neutral phospholipid to the inner layer of the membrane. The membrane thus becomes less negatively charged, allowing the Cdc42 anchor to slip out more easily. It's an example of how “a simple physical interaction can regulate the complex morphogenetic outcome observed during cell polarization,” explains Das.
Case closed? Not quite. The researchers were hopeful that they'd uncovered the mechanism based upon their observations with living cells. But in a living cell “it's hard to rule out the effects of something else,” says Li. So Das painstakingly created small bits of man-made, reconstituted membrane to test the mechanism. “It was a great challenge and required much troubleshooting before we performed it successfully,” he recalls. After a year of effort, it worked. Using an imaging technique called total internal reflection fluorescence microscopy, the team was able to show that the charge of the membrane does indeed determine how fast Cdc42 can be pulled out by Rdi1.
The next challenge will be figuring out what controls the flippase. “There are some hints that it gets even messier—that the flippase may be regulated by another kind of lipid in the membrane and perhaps other enzymes,” says Li. But the work in the new paper is already opening the door to a better understanding of the development of cell polarity in all types of cells. And it points to the power of new experimental tools and mathematical analysis that can help track and count molecules in cells, says Li.
“What the study really highlights is the future of cell biology—the ability to make these very accurate biochemical and biophysical measurements in single live cells and reconstituted systems, rather than using traditional bulk biochemical assays,” says Li.
Researchers who also contributed to the study include Brian D. Slaughter, Jay R. Unruh, William D. Bradford, Richard Alexander and Boris Rubinstein at the Stowers Institute for Medical Research.
The work was part of a project funded by the National Institute of General Medical Sciences.
About the Stowers Institute for Medical Research
The Stowers Institute for Medical Research is a non-profit, basic biomedical research organization dedicated to improving human health by studying the fundamental processes of life. Jim Stowers, founder of American Century Investments, and his wife Virginia opened the Institute in 2000. Since then, the Institute has spent over 800 million dollars in pursuit of its mission.
Currently the Institute is home to over 500 researchers and support personnel; over 20 independent research programs; and more than a dozen technology development and core facilities. Learn more about the Institute at http://www.stowers.org
Media Contact
More Information:
http://www.stowers.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Redefining Cancer Science: Rodents, Humans, and the PD-1 Puzzle
Results of a comprehensive analysis refute assumptions that a key immune checkpoint receptor functions the same in rodents and humans The Discovery of PD-1: A Milestone in Cancer Treatment Since…

Heart Surgery Risks: Low BP Linked to Postoperative Kidney Injury
First large cohort study at the Heart and Diabetes Center NRW awarded – Hilke Jung presents research project at the FoRUM conference of the Ruhr University Bochum A working group…

Microbial Secrets: Boost Plant Growth with the Power of Soil Bacteria
To stay healthy, plants balance the energy they put into growing with the amount they use to defend against harmful bacteria. The mechanisms behind this equilibrium have largely remained mysterious….



